Factors that influence the uptake of precision-guided treatment recommendations in paediatric cancer: a systematic review
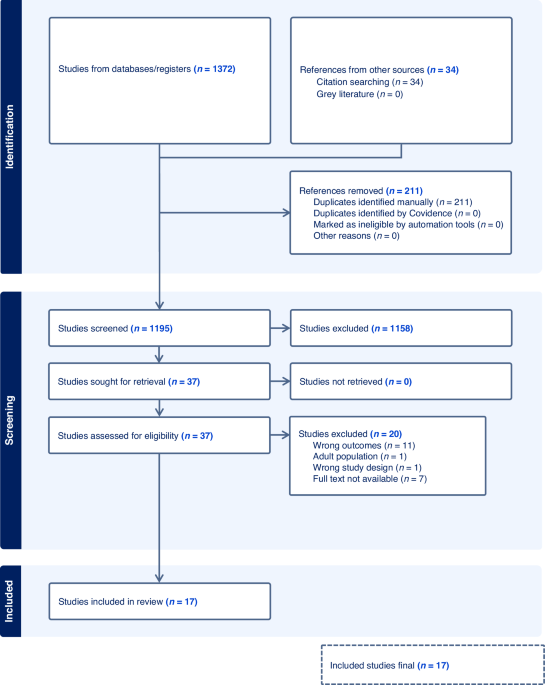
From 1372 articles returned by the search strategy, 1158 articles were excluded after title and abstract screening, and 20 articles were excluded after full text screening. This led to 17 eligible articles for which data were extracted. The study selection process is illustrated in the PRISMA flow diagram (Fig. 1). All 17 studies were of adequate-high quality based on the MMAT (Supplementary Table 1).
PRISMA flow diagram: search and selection process.
A meta-analysis was not feasible due to the heterogeneity of study design and methods used. Thus, a narrative synthesis was conducted to summarise the findings of the included studies, as shown in Table 2.
Study characteristics
All studies were conducted between 1 May 2012 and 1 April 2021. No conflicts of interest were reported among these studies.
All 17 eligible studies were conducted in developed countries, mostly in North America (9 in the USA, 1 in Canada). The remaining seven studies were led from Australia (2 studies), France (2 studies), Germany (1 multi-institutional study), Netherlands (1 study), and UK (1 study) (Table 2).
Methodologies were diverse with 15 quantitative studies [7, 14,15,16,17,18,19,20,21,22,23,24,25,26,27] and 2 qualitative studies [28, 29]. This included 13 feasibility studies, 1 cohort study and 1 Discrete Choice Experiment (DCE) survey. Among the 15 quantitative studies, 13 studies involved paediatric cancer patients and assessed the feasibility of molecular profiling and/or identifying actionable alterations and making PGT recommendations to paediatric patients with cancer. In these studies, information related to influencing factors regarding treatment decision-making was often reported briefly and lacked comprehensive detail. One cohort study reported that, with highest target priority level of recommendation, 47.6% (20 out of 42) patients took up PGT recommendation; the study also compared the clinical benefit (progression-free survival days) between patients who took up PGT recommendation and other patients [27]. The DCE survey aimed to quantify the importance of those influencing factors among healthcare professionals, parents and community members [14]. The remaining 2 qualitative studies interviewed paediatric healthcare professionals and parents to investigate the influencing factors in detail [28, 29].
Sample characteristics
A total of 3976 patients, 227 clinicians, 189 parents or caregivers, and 532 general community participated in the included studies. Healthcare professionals included paediatric oncologists, physicians, and clinicians (Table 2). The patient populations in the included studies consisted of individuals with high-risk cancers or those with relapsed or refractory disease following conventional therapy, both associated with a limited likelihood of cure (Table 2).
Among patients for whom a diagnosis was provided, 339 were diagnosed with haematological malignancies including acute lymphoblastic leukaemia (ALL)/acute myeloid leukaemia (AML)/lymphoma, 665 patients with central nervous system (CNS) tumours including high-grade glioma (HGG), medulloblastoma and ependymoma. There were 2238 patients diagnosed with extracranial solid tumours, and 57 patients with rare cancers.
Keywords and terminologies
In 17 studies, 9 studies incorporated a “keywords” section as part of their paper. Keywords and terminology employed in the included studies differed, which contributed to the heterogeneity of results.
A summary of the equivalent terminology derived from the included studies is presented in Supplementary Table 2. For example, terms of precision medicine were used interchangeably with “personalised medicine”, “targeted therapy”, and “molecular targeted therapy”; while terms to indicate molecular analysis included “next-generation sequencing”, “whole exome sequencing” among others.
Characteristics of precision medicine programmes in included studies
Treatment recommendations and tiering of supporting evidence
Ten studies [7, 16, 18, 19, 21,22,23, 25,26,27] described the use of a tiering system to rank the supporting evidence for PGT from the highest to the lowest, such as described earlier for the Boston tiering system (Tier 1, clinical evidence in the same cancer, all the way through to least evidence level, Tier 5, consensus opinion) [6]. This hierarchy of evidence underscores the varying degrees of confidence in treatment recommendations based on current scientific literature, however multiple systems with different standards can further complicate decision-making processes.
The 17 studies included five different tiering systems, each with between five to seven tiers, ranking the available evidence for a PGT from highest to lowest. Tiering systems included the Boston tiering system (iCAT) (4 studies; 5 tiers) [7, 19, 21, 27], OncoKB Clinical Actionability Tiers (6 tiers) [16, 26], Oxford Centre for Evidence Based Medicine guidelines [22], Level of evidence of eligible Gene Variants (6 tiers) [23], and Clinical priority score (7 tiers) [18, 25].
Molecular tumour board (MTB) recommendations and turnaround times
In thirteen (from 17) studies, the PGT recommendation was provided by regular MTB meetings as part of the study design, which discussed the best possible treatment plan with the molecular testing results.
Nine of the included studies reported on TAT [7, 17,18,19,20,21, 23,24,25]. The average turnaround time was 42.3 days (Table 2).
Molecular analyses performed
The types of molecular analysis performed varied among the included studies, as shown in Table 3. Among them, 8 studies conducted somatic whole exome sequencing (WES) [17,18,19,20,21,22, 25, 28]; 2 studies used low-coverage somatic whole genome sequencing (lcWGS) [18, 25]; somatic DNA panel sequencing was commonly used (n = 6 studies) [7, 16, 19, 22, 24, 27], among them 2 studies conducted targeted next generation sequencing (OncoPanel) of tumour DNA [7, 27]; and 9 studies performed RNA sequencing [7, 17,18,19,20,21, 23, 25, 26]. Four studies conducted DNA methylation profiling [18, 21, 23, 25]. Four studies performed germline whole exome sequencing (WES) [16, 19, 21, 28], and 2 studies conducted germline whole genome sequencing (WGS) [15, 26]. In one hypothetical study, a genomically guided approach was considered for treatment, as referenced in ‘You are considering recommending genomically-guided treatment’; however, no specific testing details were reported [14].
Pre-clinical drug testing
Only one study reported on functional ex vivo drug sensitivity testing, with preclinical/drug testing data discussed at the MTB when treatment recommendations were formulated [15]. None of the included studies reported on patient-derived xenograft models (PDX)-related data.
Diagnostics and PGT clinical uptake
Thirteen studies reported on the molecular results generated for 1209 patients [7, 15, 17, 19,20,21, 23,24,25,26,27] and 311 tumour samples [16, 18] with actionable alterations. Overall, 1438 patients received formal treatment recommendations which means at least 36.2% (1438/3976) of patients had PGT options. Among the 1438 patients from the 13 included studies who had PGT recommendations, 476 patients received recommended therapy, thus the uptake rate was 33.1% (476/1438). The reported PGT adoption rate of the 13 studies that provided data ranged from 7 to 46% (median 16%). The utilisation of PGT across the included studies is illustrated in Fig. 2.
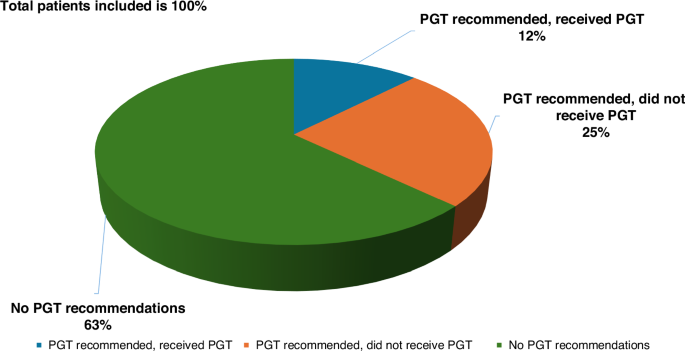
A total number of 3865 patients from the systematic review were included. Please see the reasons for not utilising the PGT recommendation in Fig. 3.
There were 1.96% patients (78/3976) whose diagnosis was changed/refined due to the molecular analysis. There were 7 studies that reported germline testing results of 10.54% (195/1849) patients with germline alterations. There was no data reported regarding the use of germline results to influence treatment recommendations and/or surveillance strategies.
Factors that influenced clinical uptake
Figure 3 presents the key factors influencing the clinical uptake of PGT, as identified across the included studies. Various factors were considered, according to the study design of included studies. For example, parents’ and oncologist perspectives of impact of genomic information on patient care were analysed [24] and themes were subsequently grouped into three domains for this systematic review: decision maker-related characteristics, decision-specific criteria, and contextual factors [30].
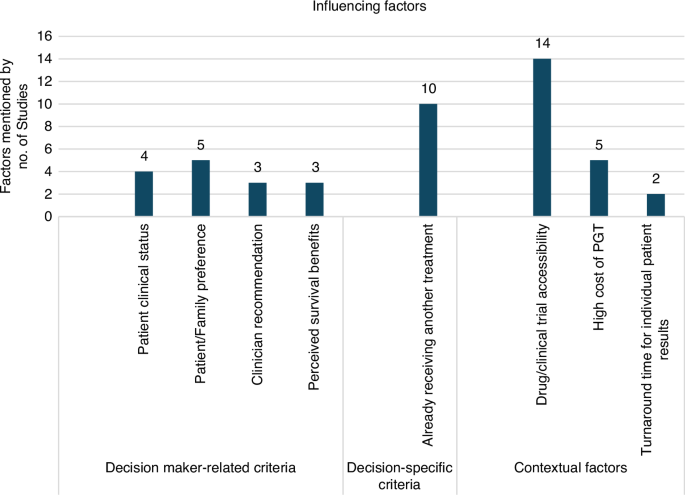
This figure depicts the most mentioned influencing factors regarding the clinical uptake of PGT within the included studies, all influencing factors were grouped by three domains.
No studies looked at which specific factor correlated with PGT uptake. Only one DCE paper ranked the factors among different populations: healthcare professionals, patient/parents, and the general community [14].
Decision maker-related criteria
Patient clinical status
Four included studies described how patient clinical status influences the decision to take up PGT. In three studies, PGTs were not taken up to due to the patient being disease-free [20, 23, 27], stable disease [23], or well controlled by other therapy [27]. On the other hand, 15% (n = 31) of patients had sufficiently poor clinical status which meant PGT was not acted upon [23]. Five patients from one study who were in poor health (8.3%) died soon after sequencing was performed [24]. Three studies explored specific information regarding patient clinical status [8, 14, 20]: from 299 patients, 71 patients (23.75%) had poor performance status due to progression of underlying malignant disease, and 13 patients (4.35%) had poor performance status due to toxicity from previous treatment(s) [25]. Two studies reported clinical deterioration of the patient [16], advanced disease or death (3 patients, 16%) [27] as reasons not to pursue PGT.
Patient/Family preference
Five studies reported that patient/family preference affected the likelihood of PGT adoption in two directions [20, 24,25,26, 29]. One study described that the family/patient’s support or preference for the PGT recommendation could increase the likelihood of uptake [29]. Four studies reported that patient and/or parent preferences were important in the decision not to take up the PGT recommendation: in 2 studies, 25 patient/guardians [25], and 1 patient (1/25, 4%) did not pursue PGT due to patient/family refusal [26]; and 1 patient (1/21, 4.76%) refused PGT because of family wishes [24]. One study mentioned both family and physician preference as one of the reasons for patients not taking up (or acting on) PGT recommendations [20].
Clinician recommendation
Three studies offered additional insights into influencing factors for PGT uptake, related to clinician recommendation. Five patients (5/21, 23.8%) were given conventional therapy with chemotherapy or a surgical intervention as determined by their physicians [24]. Some patients did not accept PGT simply because their treating physicians thought no additional therapy was necessary [20]. Similarly, the only DCE study reported that whether the health care professional recommended the treatment to the patients affected the PGT adoption [29].
Patient – doctor communication barriers
One qualitative study investigated perspectives from treating physicians and parents [28]. It addressed treating physicians’ concerns that communicating complex findings from WES with parents might be disruptive during the decision-making process. This was based on the assumption that complex findings might lead to misinterpretation of the relevance of some findings, which could distract parents from making decisions in terms of cancer treatment and care for their child [28]. Despite this assumption, this study demonstrated that parents of paediatric patients with solid tumours who enroled in clinical trials have a strong desire to learn and do everything they could to support their child. Maximal provision of information was helpful for parents to make an informed decision and to take advantage of this novel technology [28].
Altered patient quality of life (QoL) under treatment
Two studies reported patient’s quality of life (QoL) or possible QoL after implementing the PGT recommendation as an influencing factor of uptake [14, 29].
Perceived survival benefits
Three studies reported that clinician perceived survival benefits [14], the severity of the patient condition without a genomic approach [29], or magnitude of the possible clinical benefits positively increase the likelihood of PGT adoption [15]. Additionally, McCarthy et al reported that the greater the perceived benefit to the patient, the more likely the clinician is to recommend PGT [29].
Decision-specific criteria
Already receiving another treatment
Four studies reported receiving standard treatment as one of the reasons that patients did not implement PGT recommendation. These four studies referred to standard treatment, conventional treatment, first-line therapy, proven standard relapse therapies, and frontline treatment, which were distinguished from clinical trials [7, 16, 24, 25]. In one study, 200 patients (48%) were not expected to consider PGT as they were newly diagnosed, indicating that standard treatment was the preferred option. Despite experiencing relapse/refractory disease, only 4 (7%) out of 57 patients took up PGT recommendations, with a considerable proportion of patients (no specific number given here) receiving conventional therapy after they underwent sequencing [16].
Six studies reported patients were already receiving other alternative treatment [3, 9,10,11, 18, 22] as a reason to not take up PGTs. One quantitative study reported that the most common reason for not adopting PGT was receipt of other treatment in 32% (64/200) patients in their study [23]. Two studies mentioned that PGTs were saved for a later time point as alternate treatment [16, 17].
Other alternative treatment here means therapies including no cancer-directed systemic therapy for 49/204 (24%) patients, enrolment on a phase I/II clinical trial which did not require biomarker sequencing [16], attempting third-line therapy (other non-PGT experimental therapy) [27], or other alternative therapies with no further information [15, 17, 23]. One quantitative study reported that an alternative regimen was chosen for 14.67% (11/75) of patients [17].
Turnaround time for individual patient results
Two studies reported that patients did not have a chance to implement PGT due to the turnaround time for sequencing results and a treatment recommendation. Some patients died before finalisation of the molecular analysis and molecular tumour board report (n = 22, 7.36%) [25] or were in palliative care due to the testing results being generated too late (mean TAT = 54 days, range, 15-114 days) in the clinical course [20].
Tier level of supporting evidence
Ten (out of 17) studies included tiering systems regarding level of supporting evidence for treatment recommendations [7, 16, 18, 19, 21,22,23, 25,26,27]. Only one study reported that patients who received the highest level of recommendation, referred to as a Tier 1 recommendation in the iCat recommendation tiering system, were more likely to adopt PGT when compared to patients who did not receive a top-tier recommendation [7]. The rest of the 9 studies did not provide this information related to treatment decision-making.
Contextual factors
Drug/clinical trial accessibility
Fourteen out of 17 included studies (82.4%) reported that the accessibility of treatment and/or drugs was a significant issue influencing uptake of PGTs.
Five studies described patient-related factors and/or clinical trial eligibility that can affect access to drug treatment. In some cases, patients were not eligible to adopt the PGT due to clinical trials restrictions [22], persistent metabolic disorders [24] or inappropriate current clinical status [27]. Harttrampf et al. [17]. reported 13 patients (30.95%) had no access to a clinical trial because of age, timing of trial opening, no respective age slot available, or trial inclusion criteria not fulfilled (i.e. patient not eligible for the trial).
Six studies reported that PGT uptake was hindered by lack of an available drug or clinical trial [7, 16, 20, 24,25,26]. Two studies highlighted that numerous new drugs have not yet been approved for use in children due to insufficient data on their effectiveness in treating paediatric diseases or their safety in children [17, 21]. Furthermore, the process of obtaining experimental drugs that are not approved by the FDA (or relevant national drug approval system) for paediatric patients is time-consuming, and access to these drugs is rarely granted to younger patients [19, 21]. Experimental medicines being tested in clinical trials or newly licensed drugs for adults are rarely given to young patients, even in cases when compassionate use is considered [21]. For 11 (out of 299, 3.68%) patients, the drug formulation was unavailable despite the targets being identified [25].
High cost of recommended treatments
Two studies described cost as a major barrier to the implementation of PGT, despite the reduced cost of sequencing [25], as the personnel cost of precision medicine is expected to remain high [19].
Once a PGT recommendation is made, the cost of drug was also a barrier to access, as described in five studies [14, 16, 21, 25, 29]. Five studies reported cost-related factors that ranged from (total) cost of treatment [14, 29], insurance company declined cost coverage [25], and the off-label use of high-cost agents not covered by insurance companies [14, 21, 29]. These factors can all directly impact the adoption of PGT [14, 29]. Patients did not adopt PGT due to difficulties accessing novel drugs on a compassionate-use basis, which is cost-related, with no specific patient numbers provided [16].
link






