Interplay between genes and social environment: from epigenetics to precision medicine
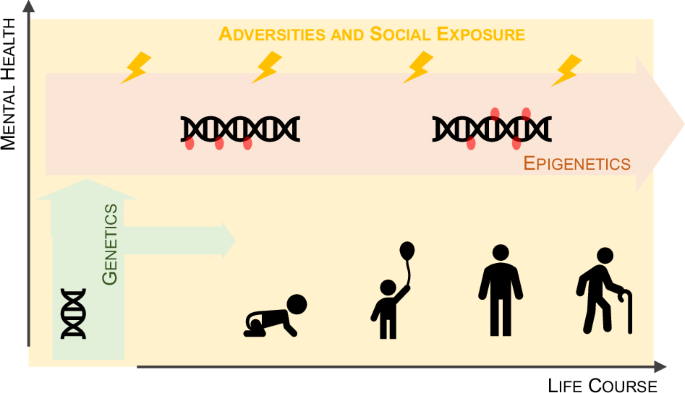
Epigenetics refers to the study of how environmental factors can alter gene expression without changing the underlying DNA sequence. These changes can, in turn, influence behaviour, cognition, and personality. The Diathesis/Stress model, introduced by Zubin and Spring in 1977 [6], offers a framework for understanding mental health disorders. According to this model, such conditions arise from the interaction between genetic vulnerabilities (diatheses) and environmental stressors. It suggests that individuals with a genetic predisposition to mental illness may require specific environmental triggers, such as trauma or adversity, to manifest the disorder. This model emphasizes that mental health disorders result not from genetics or environmental factors alone, but from the interplay between the two.
A twin study by Kendler and colleagues [7] highlighted that 37% of the variance in susceptibility to major depression could be attributed to genetic factors, with substantial overlap between the genetic factors influencing depression and other psychiatric disorders. Similarly, a meta-analysis by Hettema and colleagues [8] showed that between 30% and 50% of the variance in anxiety disorders could be explained by genetic predisposition, with variations depending on the specific disorder. These findings underscore the significant role of genetics in mental health. However, environmental factors also contribute to mental health conditions. A more recent adoption study revealed a significant resemblance in the prevalence of major depressive disorder between children and their adoptive families, indicating environmental transmission [7]. Even if children are not genetically related to their adoptive parents, being raised in a household where caregivers, such as step-parents, struggle with depression increases the likelihood of the child developing depression themselves. This supports the idea that environmental factors, such as the mental health of caregivers, can shape an individual’s vulnerability to mental illness.
Changes in gene expression within limbic brain have been associated to aberrant epigenetic regulations with potential causative roles in development of depression and stress-related disorders, such as post-traumatic stress disorder and various anxiety disorders [9]. Consistently, antidepressant medications may exert, at least in part, enduring therapeutic effects by being mediated through epigenetic mechanisms [9].
The environmental effects were also shown to be transmissible with mechanisms of epigenetic inheritance. In mice, chronic exposure to psychosocial stress led to a significant decrease in 5-methylcytosine levels in germ cells. Specifically, psychosocial stress appeared to alter DNA methylation in gene regulatory regions that are involved in transcriptional regulation. These findings suggest that chronic stress disrupts the male reproductive system by inducing abnormal epigenetic changes in male germ cells [10]. This indicates how chronic stress negatively affects germ cell development in the testis and that these effects can be inherited by offspring.
Behavioural traits in mammals have been linked to non-genetic transgenerational inheritance involving the germ line, with epigenetic modifications and non-coding RNAs in germ cells playing a key role [11]. These mechanisms appear similar to non-genetic transgenerational inheritance observed in parental exposure to toxicants or metabolic stress, whose offsprings display higher risk of deficits [12,13,14].
Consistent with epigenetic research in this area [15], development of common mental health disorders appears to be the integration of multiple influences by an interaction between genetic predispositions and environmental factors, including socioeconomic status, prior trauma, and the mental health of caregivers.
link






