National variation in the treatment of lung cancer in a universal healthcare context
Over our study period, the most common treatment received by those with lung cancer in Aotearoa New Zealand was radiation therapy, either in isolation or combined with systemic therapy (combined total: crude 37%). This trend held when adjusting for multiple factors, including stage of disease and comorbidity (fully-adj. 40%). Systemic therapy was the next most common treatment modality, used in around a third of the cohort (crude 28%, fully-adj. 33%). Surgery was the least common treatment modality, used in around 15% of the cohort (crude 18%, fully-adj. 15%).
Our observations regarding variation in treatment patterns by population sub-group are detailed below.
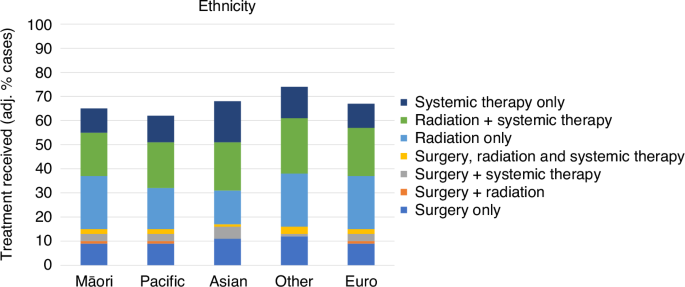
Ethnicity
We observed that, once adjusted for all other covariates, Māori did not appear to be receiving a different pattern of lung cancer treatment compared to Europeans. At a glance, this suggests that the combination of treatment received by Māori with lung cancer is unlikely to be a contributor to enduring disparities in cancer survival for Māori. However, this finding must be considered alongside both our recent observation of lower surgical rates among Māori with lung cancer, where Māori were less likely to receive surgery (age-standardised %: Māori 14%, European 20%) including curative surgery (Māori 10%, European 16%) [8], as well as our observation in the current study that Māori were less likely to access surgery only before adjusting for covariates other than age (age-standardised %, combined surgery totals: Māori 15%, European 21%). In combination, these findings suggest that Māori are indeed less likely to receive surgery than Europeans, but that this difference is largely explained by differences between these groups in factors such as comorbidity and stage (Supplementary Material 2). The findings of the current study suggest that once these factors are accounted for, Māori receive largely the same pattern of lung cancer care as Europeans; but this should not be misinterpreted as meaning that there is no difference between Māori and Europeans in terms of received treatment. Our findings merely suggest that it there is no apparent impact of Māori ethnicity on pattern of care independent of factors such as stage and comorbidity.
We found that Pacific peoples appeared to be marginally less likely to access radiation therapy than other ethnic groups, with no commensurate increase in receipt of other forms of treatment—even after adjusting for plausible drivers of this deficit in treatment, including comorbidity and stage of disease. This appeared to be the primary contributor to the overall slightly lower rates of lung cancer treatment among Pacific peoples in the adjusted results. This slightly-reduced access to radiation therapy will not be related to geographic access to care, because we have already shown that Pacific peoples with lung cancer almost exclusively live within urban hubs with radiation treatment facilities [21]. While further work regarding this observation is warranted, we note that relatively low numbers of Pacific patients means that these findings may be imprecise, and therefore any differences should be interpreted with caution.
We noted that Asian peoples appear to be accessing more lung cancer surgery and less radiation therapy than other ethnic groups, which is partly explained by a tendency toward earlier stage of disease at diagnosis and less comorbidity among this cohort. A greater proportion of Asian patients with lung cancer are non-smokers [22]: in New Zealand, a previous study found that 35% of Asian patients with lung cancer were non-smokers compared to 8% of European patients and 1% of Māori patients [23]. This may lead to an increased propensity toward low-grade adenocarcinomas among Asian patients that should be amenable to surgery, and the patients were relatively young and fit enough to receive this treatment (around 70% of Asian patients in our cohort had a comorbidity score of ≤0; Supplementary Table 1).
Stage
In terms of stage of disease, we found that surgery receipt goes down with increasing stage of disease, while the relative importance of systemic therapy and radiation therapy increases with increasing stage. Improving access to early detection of lung cancer through interventions including screening [24] would change this pattern by increasing the viability of surgery as a curative treatment option.
We observed an unexpected near-absence of radiation therapy only among those with Local stage. We expected to see more instances of stereotactic ablation radiation therapy (SABR) among this cohort, as SABR became a common treatment for early-stage lung cancer over our study period [25]. Within our own cohort, we found that use of SABR occurred in <1% of those with Local, Regional or Advanced disease, and 4% of those with Unstaged disease (crude %; data not shown). Recent data from a large clinical registry found that SABR was used to treat around 11% of early-stage lung cancer (TNM Stage I–II) cancers [26], reinforcing the unusually low proportion treated with SABR in the current study. The first plausible reason for this observation is that SABR was under-used in the New Zealand context over this study period, although there is limited evidence to suggest that this was the case [25]. A second plausible reason is that patients who receive SABR might not be registered on the NZCR, because the primary source of data for this registry is pathology records [10], and early-stage patients with lung cancer who receive SABR may be less likely to have a pathology sample taken as part of their care. A recent granular analysis from the same clinical registry found that a small proportion (6%) of their clinical cohort were not registered on the NZCR, and that around a third (29%) of this group had Stage I disease [27]. This observation, in addition to the near-absence of radiation therapy among those with early disease in our cohort, suggests (a) that we are likely missing a small proportion of cases from our cohort, (b) that this absence is not likely to be random but rather patterned by stage, with many of these missing registrations likely to be of Local stage, and (c) this absence precludes us from fully describing the pattern of treatment received by those with Local disease.
Rurality
Contrary to expectations, rurality appears to have no impact on the combination of treatment received by lung cancer patients in Aotearoa, both before and after adjustment for possible confounding and mediating factors such as deprivation. This suggests that those living in rural areas are accessing the same patterns of care as those living in urban centres, despite having to overcome barriers to accessing surgery and radiation therapy which are almost exclusively delivered in our major cities [28]. This observation does not negate the need for us to decentralise care and provide it close to where people live wherever possible—particularly since treatment centralisation tends to make access easier for some population groups over others (e.g. odds ratio of Māori with lung cancer living ≥200 km away from radiation therapy: 1.41, 95% CI 1.25–1.60) [21].
Deprivation
Increasing deprivation appeared to slightly reduce both the overall proportion of the cohort who received treatment as well as the proportion who received surgery only. While the difference in surgery rates was greatest prior to marginal standardisation for other covariates, a small disparity did remain even after adjustment for these factors. This reinforces the likelihood that deprivation can act as a barrier to treatment access independent of factors such as rurality, as well as the importance of considering the independent impact of deprivation on outcomes within equity-focussed analysis (e.g. ethnicity comparisons).
Comorbidity
The overall proportion of treatment reduces with increasing comorbidity, with this deficit apparently driven by a reduction in receipt of systemic therapy (but not other treatments) as comorbidity increases. On the one hand, this observation is surprising. Previous research (including our own) has shown that the presence of comorbidity reduces the likelihood that a person with cancer will receive surgery; it appears that this impact does not apply to the lung cancer cohort, which may be explained by the heightened presence of comorbidity among those with lung cancer relative to other cancers [29]. (In other words, much of our lung cohort have comorbidity, so there is less opportunity for observing a strong relationship with treatment receipt.) On the other hand, despite the heightened morbidity within the cohort, we still observed a strong relationship between increasing comorbidity burden and likelihood of receiving systemic therapy only (C3 category ≤0 vs >2, adj. 11% vs. 8% respectively, adj. OR 0.68, 95% CI 0.58–0.79) and radiation therapy plus systemic therapy (21% vs. 13%, adj. OR 0.49, 0.43–0.57). In order to assess the extent to which the latter was driven primarily by systemic therapy access (as indicated by the systemic therapy only finding), or whether radiation therapy access was also a factor, we examined the receipt of any radiation therapy (regardless of combination with other treatment) by comorbidity status, and found that those with a C3 score of >2 were less likely to access any radiation therapy compared to those with a C3 score of ≤0 (adj. 46% vs. adj. 38%, adj. OR 0.71, 95% CI 0.65–0.78), despite there being no difference between these comorbidity groups in terms of access to radiation therapy only within the current study (both 22%). In combination, these findings suggest that comorbidity is influencing both the receipt of radiation therapy and systemic therapy, rather than surgery, independently from other factors including stage of disease (to the extent to which we can adjust for this factor with our current data). Further nuanced investigation of this trend with clinical registry data (and complete TNM staging and ECOG performance status data) will add granularity to this observation; however, in the meantime, our findings suggest that comorbidity is an important determinant of access to radiation therapy and systemic therapy in the lung cancer context, and as such the management of comorbidity among this population could improve access to this treatment, with subsequent improvement in outcomes.
Recommendations
Based on the findings outlined above, we make the following recommendations regarding system- and clinical-level changes arising from this study:
-
Patterns of treatment depend on stage of disease, and as such, a population shift toward early-detection of lung cancer would result in a corresponding shift away from the patterns of treatment observed for late stage disease (proportionally more systemic therapy) to those observed for early stage disease (proportionally more surgery). A lung cancer screening programme is one means by which such a shift could occur [24].
-
Comorbidity independently influences access to radiation therapy and systemic therapy, and therefore, the primary prevention of morbidities including diabetes, heart disease and others would serve to improve access to these cancer therapies among those with lung cancer. In addition, management of comorbidity and prehabilitation activities could also improve treatment access—with such an improvement set to disproportionately improve access for those populations with an inequitable burden of comorbidity (such as Māori and Pacific).
-
Pacific peoples appear to not be accessing radiation therapy to the same extent as other ethnic groups, even after adjusting for factors including comorbidity. Further work is required to understand the potential barriers that Pacific peoples may uniquely face in accessing this care.
-
Rurality does not seem to influence either the pattern of lung cancer treatment received or the overall likelihood of treatment—but that does not mean that our rural populations are not experiencing barriers to care. Rather, it suggests that these barriers are being overcome by a resilient rural population, but since much of lung cancer treatment is delivered in central hubs [21], this resilience will come at a price. Any and all activities that aim to reduce rural barriers to cancer care access, including improvement in the availability of up-front travel expense funding through transport assistance programmes, are sorely needed in acknowledgement of the systematic problem that centralisation of treatment creates for those diagnosed with cancer while living in rural communities.
Strengths and limitations
A key strength of this study is the use of national-level data, maximising the generalisability of our findings to the lung cancer population within New Zealand. As noted above, it is likely that we are missing a small number of the total incident lung cancers in New Zealand over the study period, and while this likely undercounts receipt of radiation therapy among our cohort (as outlined above), this should not meaningfully impact the key themes of our observations. We noted above the limitations of the staging information currently available at a national level for lung cancer in New Zealand; this limits the granularity of our observations, and reinforces the importance of rapid improvement in the availability and linkage of clinical staging to national health records, with work currently underway in New Zealand to address this gap [30]. Finally, we note that since our study cohort ceased at the end of 2019, there may have been some changes to treatment modality since this time, such as increased use of immunotherapy and targeted therapy in the treatment of more advanced disease.
link






