The relationship between uncertainty and trust in genomic medicine and research: A literature review and thematic analysis
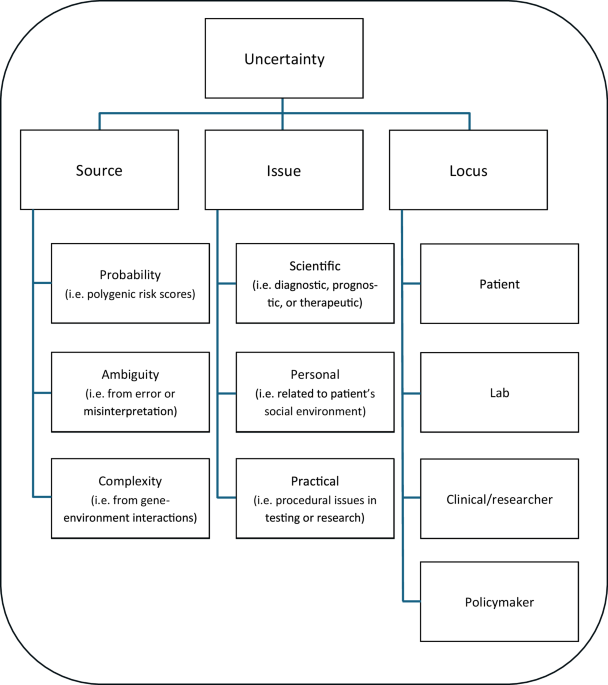
The concepts of uncertainty and trust, as well as the contexts in which they are applied, are inconsistently employed across the literature. Notably, the uses of uncertainty and trust in the publications reviewed here are largely reflective of Han et al.’s taxonomy of medical uncertainty [4]. Complexity, probability and ambiguity (their three identified sources of uncertainty) were very common forms of informational uncertainty, with each category appearing in between 23% and 50% of reviewed studies. Similarly, the three ‘issues of uncertainty’ they propose – scientific uncertainty, practical uncertainty, and personal uncertainty – were common themes in our review, although we have opted to categorise practical uncertainties as being ‘processual’ uncertainties because our subject matter extended beyond the context of ‘structures and processes of care’ [4] and into the context of data management, as noted by the code ‘future uses of data’. While studies such as Han et al.’s have addressed the possible categorisations of inconsistent uses of the concept of uncertainty, no such method has yet been applied to the concept of ‘trust’ in medicine.
Across the articles we reviewed, the interaction between trust and uncertainty has only been outlined in a handful of studies; only three of the studies identified in this review make causal claims about the relationship between trust and uncertainty. What these few studies suggest is that there are plausible causal relationships between trust and uncertainty in both directions, depending on context. Bartley et al. claim that patient trust in their healthcare providers can have an effect on the likelihood with which they perceive uncertainty in genomic test results [22]. In the other direction, Gille and Brall claim that uncertainty about future use of genomic data can impact the trust that people have in biobanks [25], while Horton and Lucassen claim that scientific uncertainties in population studies can undermine general confidence in the predictive abilities of genomics [29]. Notably, the different relational dynamics within clinical and research settings may help to account for some of the differences in focus of these studies: while Bartley et al.’s clinical context paper is concerned with personal uncertainty experienced by patients and factors impacting those experiences, Gille and Brall explore the broader societal values shaping donor trust and biobank participation. Despite these domain-specific differences, the vast majority of surveyed studies described coincidental and mediated relationships, highlighting complex interdependencies between these trust and uncertainty.
To characterise these relationships in a way that renders them of value to our research question, we propose that trust and uncertainty can be categorised by the role that uncertainty plays in offering an opportunity for the actor to demonstrate their trustworthiness. There have been calls for clinicians to accept and engage with patient’s experiences of feeling uncertain and encountering uncertain information, as opposed to focusing on how both kinds of uncertainties can be lessened [6, 8, 9]. The utilisation of mechanisms to reduce uncertainty or to minimise the subjective or disorienting experience of uncertainty – such as the normalisation of uncertain results through counselling, or through reclassification of uncertain results through additional research – is also consistently illustrated [43, 44]. Still, the need to acknowledge and adapt for uncertainty in interactions suggests some genetic counselling approaches help build trust. For example, a ‘counselling approach’, which is focused on providing information in ways that work for the patient (e.g., according to their background, understandings and perceptions), and generating rapport and evidence of empathy, could allow for uncertainty to be better navigated, and for trust to be built [45].
The benefits of altering, or adopting new processes and practices to deal with uncertainty are similarly being highlighted. In a review of guidelines concerning the management of uncertainty in prenatal genomics, Klapwijk et al. identify strategies that are engaged in relation to different kinds of clinical/scientific uncertainty [43]. For instance, the authors suggest that involving a multi-disciplinary team (i.e., different professional staff, including laboratory technicians and clinicians) in decision-making around testing and reporting may work to reduce uncertainty. In fact, when it comes to reporting, or not reporting VUS, input from additional stakeholders such as other laboratories, and/or relevant multi-disciplinary committees, is recommended. Meanwhile, others have drawn attention to the need for genomic testing to foster values and qualities such as resilience, welfare, autonomy and solidarity to deal with the cognitive fact of uncertain situations [9].
Focus is additionally placed on the types of strategies that can equip clinicians to better assist patients in managing or accepting personal uncertainty [8, 46]. Key to this is ‘patient-centred’ communication [5], which recognises that both too little and too much information can lead to, or exacerbate, personal uncertainty [5, 12]. The perception of poor patient communication has been suggested to correlate with the reporting of mistrust in healthcare practitioners [45].
Within the context of genetic counselling, emphasis on patient-centred communication can again be understood to promote the use of the counselling approach as described by Kessler [45]. While alternate approaches such as the teaching model centre on information delivery, the counselling approach accounts for personal uncertainty by giving attention to not only the patient’s specific communication requirements, but the outcomes they are seeking from appointments more generally. At the same time, some literature suggests that the timing of information delivery may be significant when it comes to minimising uncertainty. For example, Klapwijk et al. note the importance of preparing patients in pre-test genetic counselling appointments, and via related consent documentation, by explaining the level of uncertainty that could, or that is likely to characterise, or result from, genetic tests. Moreover, the authors signal the benefits of drawing attention to the control patients have in the process, with patients’ ability to opt-out of receiving secondary or incidental findings highlighted. This is perhaps why the development of guidelines that account for the views of all key stakeholders – including, patients – is being promoted to ease experiences of uncertainty in prenatal genomics [43].
Still, because the uncertainties described in studies are often a common or necessary feature of genomics, instead of attempting to minimise uncertainty, it may be necessary to focus on adapting and responding to the events and information that contribute to it. Evidently, reflecting on clinical processes, practices, approaches and information delivery is likely to be beneficial when it comes to navigating both uncertainty and trust in genomic medicine and research. Still, following Newson et al., we argue that an ‘ethics of genomic uncertainty’ is needed to foster trust in the presence of the kinds of uncertain information and situations identified in this review [9]. This may involve clinicians engaging with core ethical concepts such as resilience, welfare, autonomy and solidarity [47, 48], and developing methods for addressing and actioning such in their interactions with patients. For instance, in addition to making patients aware of uncertainties that may arise, offering recommendations and support in relation to how patients can deal with the uncertainty, may be beneficial.
The most common form of informational uncertainty identified in this study, risk and probability, presents a typical case of irreducible uncertainty in both clinical and research genomics. Studies that describe probability or risk-based uncertainties often describe these in negative terms because of the perceived consequences for a patient’s behaviour or emotions: i.e. feeling anxious about genomic test results [12, 24], being reluctant to make healthcare decisions based on risk [23], or becoming overwhelmed by numerical risk information [13]. Similarly, the forms of clinical and scientific uncertainty identified in this review often concern the ordinary functioning of scientific and clinical practice, such as disputes in the interpretation of test results, debate about the value of genomic testing, classification of genetic variants as being of uncertain significance, variant reclassification, and uncertainties surrounding ever developing scientific practices. While communications practices may influence how patients recall or respond to probabilistic information or topics of medical debate [22], it is not clear that the elimination of their awareness of such uncertainties will promote trust, let alone be worthy of trust.
While some of the reviewed studies posit that reducing personal uncertainty should be a goal of genetic counselling and good communication between specialists and laypersons, most studies acknowledge that the relationship between uncertain situations and trust is complex or at least mediated by an intervening variable. Most common among the mediating variables described are the work and strategies that genetic counsellors, researchers, and other public-facing persons use to communicate genomic test results. In such studies, communications strategies often mediate how uncertain outcomes shape trust, such that uncertainty can be a necessary precondition to building trust. In McMahon et al.’s study, for example, the use of appropriate communications strategies could work to foster trust between researchers and study participants in that uncertainty provides an opportunity to demonstrate trustworthiness.
It is also worth noting here the absence of research that socially contextualises how uncertainty shapes trust between communities (such as migrant, sexually or gender diverse, or Aboriginal and Torres Strait Islander) and data custodians. While a single study observed that ‘DNA biobanks and biorepositories need to engage more successfully with indigenous [sic] communities’ [35] and two further studies described the need for genomics to engage further with ‘ethnically diverse’ groups [16, 36], none described how genomic uncertainty might be related to trust for peoples in these social contexts. Addressing this literature gap will require the recognition that some forms of genomic uncertainty are precursors to trust between patients/participants and data sponsors (i.e. health services and biobanks). Patients and research participants, although ideally ‘informed’ of their decisions, will necessarily rely on the advice given and promises made by genomic data custodians and their representatives in the presence of uncertain outcomes. Thus, we may describe some forms of uncertainty as ‘productive’ of trusting relationships: a genetic counsellor need not promise certainty to build a trusting relationship with a patient; just as a biobank need not describe every possible research use of genomic data to inspire the trust of donors. Rather, both are trusted to act, to some degree, in the interests of patients and donors. In the case that uncertainty becomes a mechanism for developing trust, we describe these forms of uncertainty as ‘productive uncertainties’.
The case of First Nations peoples is illustrative here. The experiences that First Nation peoples have had, and continue to have, in relation to genomic research and health care, have sown distrust in the field [49]. In Australia, ‘negative past experiences with researchers and other institutional structures’ [50] are considered key to the suspicion and distrust many Aboriginal and Torres Strait Islander peoples feel towards genomics [51]. Distrust of research and medical institutions have also been reported among other First Nations peoples, across North America [52, 53], as well as racial minorities in those contexts [1, 54]. Literature exploring the health inequalities experienced by Indigenous Australians has identified collective vulnerabilities, such as the risk of unauthorised usage of historical DNA samples, uses of genomics to develop genetic definitions of Aboriginality, and uses of genetic analysis that may promote genetic discrimination [55]. The National Centre for Indigenous Genomics, which was formed in 2017 and has an Indigenous-led governance model and Board, is among initiatives to address inequitable healthcare outcomes and uses of genomic data [56]. In developing a model of data governance which requires an Indigenous-led Board to decide how to implement the wishes of data donors, the Centre utilises productive uncertainties. However, such inequities may not be fully accounted for without investigation of how genomic uncertainty may be shaping the participation of Indigenous communities in biobanking, research and healthcare.
Limitations
This study is limited by the practicalities of conducting a literature review in a field in which technologies, laws and public expectations can transform quickly due to political and economic interest in genomics. While the laws, regulations and sentiments described here may shift with media and political cycles, the more general analysis of the relationship between genomic uncertainty and trust is relevant, if temporally limited.
Additionally, the search terms used in this review reflect the disciplinary and scholarly experiences of the research team at the time of development of the review. However, given the variety of claims already reported here, it does not seem likely that this would have substantially added to our analysis.
Implications for research and practice
The outcomes of this study have implications for practice and research. Firstly, there is evidence that attention is being paid to the types of uncertainty we identified – specifically, clinical/scientific, informational, and personal uncertainty – with multi-disciplinary input throughout testing and reporting, data sharing, and transparency regarding uncertainty, being emphasised [43]. Still, based on the literature reviewed, processual uncertainty has seemingly received less focus. Additionally, some of the forms of genomic uncertainty described in reviewed studies seem likely irreducible and necessary features of current genomic science. As such, practitioners seeking to foster trust with patients, research participants or donors of genomic samples should not aim to diminish the uncertainties in the eyes of those consenting to genomic testing, perhaps leading to what Corrigan described as an ‘empty ethics’ [57]. Such an approach would not be consistent with common understandings of informed consent. Rather, following Newson’s lead here, it would seem both more prudent and ethically sound to build an ethics of uncertainty into clinical and research practices [9]. In accordance with Newson and Ormond’s articulation of this concept [47], an ethics of uncertainty could be incorporated into genomic counselling and briefings for research participants by drawing on four other core ethical concepts:
-
Encouraging resilience, through recognition of the impact of uncertainty on those affected and the need to give them time to process and recalibrate;
-
Promoting welfare, through finding an appropriate balance between too little and too much space for discussing uncertainty;
-
Recognising autonomy, through provision of a constructive and honest appraisal of uncertainty; and
-
Building solidarity, through mutual support and partnership.
In the studies reviewed, it is often the case that uncertainty is perceived to correlate negatively with trust in healthcare providers where those uncertainties are not realistically communicated or come as a surprise to patients [21, 25, 29, 32, 42]. The gap between patients’ expectations and counsellors’ desires to retain the confidence of their patients is sometimes described as the result of poor ‘communication strategies’ [42] but seems to also possibly derive from routine uncertainties (such as changing information that may result from variant reclassification) that are beyond the counsellors’ control [27]. In both instances, informing participants about the likelihood of uncertainty persisting and being part of genomic testing – in both pre and post-test appointments [43] – is more conducive to generating ‘productive uncertainties’ than attempting to minimise a patient’s awareness of uncertainty. Furthermore, in addition to focusing on the information being delivered, attention could be given to the way information delivery occurs, with patient-focused approaches – such as the counselling approach [45] – perhaps better equipped to account for the informational and personal uncertainty that can arise in genetic counselling appointments, than others. How to strike a balance between adequately informing patients, and not overburdening them, is beyond the scope of the reviewed studies, but is a worthy task for future research.
A second implication for future research on trust and uncertainty is that studies should be expanded to consider the place of social context and group membership in how genomic uncertainty impacts trust. As noted above, only three studies in this review described Indigenous and ethnically diverse groups, while none described how social group membership might impact on any relationship between trust and uncertainty. Collaborative genetic research projects with Aboriginal communities ‘depend upon strong relationships of trust between the researchers and the communities’ [51], a key step towards enabling Aboriginal communities to benefit from research while minimising harm.
While the participation of Indigenous Peoples and use of Indigenous-led governance have been described as significant steps towards building trust and achieving more just outcomes for communities [50], how uncertainty might shape these practices is not clear. Are the relationships between researchers and communities made more challenging through acknowledgement of uncertainty, or do researchers exercise an ‘ethics of uncertainty’ to build trust? How do Indigenous-led governance groups navigate the uncertainties of genomics in representing the interests of Indigenous individuals and communities? A more direct investigation of trust and uncertainty in these contexts would answer such questions.
link






